FDA Stands By Approval of OxyContin
OxyContin is one of the most highly scrutinized medications in American history. OxyContin has been an FDA-approved prescription medication for over twenty-seven years, and remains one today. FDA has reviewed OxyContin multiple times over the last quarter-century – each by different review teams under different political administrations – and continues to stand by the medication’s benefits to chronic pain patients.
FDA Is Government Health Authority Responsible for Balance Between Safety & Effectiveness When Approving Medicines for Sale in U.S.
“The FDA knew about the risks of opioids; that is precisely why the FDA designated opioids as Schedule II drugs. The FDA continues to approve these drugs for use where medically appropriate.”
Superior Court of the state of california, santa clara, November 2021[1]
FDA Repeatedly & Consistently Stands By Approval of OxyContin
1995
FDA approves OxyContin as “safe and effective” for patients with moderate to severe chronic pain following six clinical trials by Purdue. (OxyContin was never indicated for short-term pain such as from broken bones or immediately after surgery.)
- OxyContin label has always disclosed the risk of addiction
2001
Purdue works with FDA to add Black Box Warning and notify physicians of label changes
2002
FDA says “benefits of OxyContin outweigh its risks”
“FDA continues to believe that the benefits of OxyContin outweigh its risks when the drug is used according to the approved labeling.”
Dr. John Jenkins, Director of FDA Office Of New Drugs, Center For Drug Evaluation And Research, February 2002[2]
2008
FDA denies Connecticut Attorney General Richard Blumenthal’s “citizen petition” for Purdue to further revise OxyContin label
“Since 1995 when OxyContin was approved, the FDA has monitored and evaluated the safety concerns associated with OxyContin and has held meetings with Purdue, Congress, other agencies, and convened Advisory Committees regarding such issues. We will continue to closely monitor the safety of OxyContin and take actions as we believe appropriate.”
Janet Woodcock, M.D., Director, Center For Drug Evaluation & Research At FDA, September 2008[3]
2010
FDA approves abuse-deterrent formulation (ADF) of OxyContin in comprehensive review of medication (Purdue had begun investing in research in the early 2000s). By 2011, Purdue had replaced all tablets sold in the US with the abuse-deterrent formulation.[4]
2013
- FDA approves abuse-deterrent labeling for reformulated OxyContin after extensive review of studies created post-introduction of the ADF[5]
- FDA rejects request from Physicians for Responsible Opioid Prescribing (“PROP”) to implement maximum daily dose for physicians to prescribe, because:
- “… the scientific literature does not support establishing a maximum recommended daily dose of 100 mg MED.” – September 2013[6]
JANUARY 2020
FDA Director of Center for Drug Evaluation and Research Janet Woodcock sends letter to U.S. Senate asserting propriety of OxyContin approval process[7]
september 2020
FDA review team finds reformulated OxyContin reduced abuse by non-oral routes, and confirms the ADF did not lead to increased heroin use
“These postmarketing studies demonstrate that the abuse-deterrent properties of OxyContin have had the predicted effects on abuse and provide an incremental improvement over the original formulation that did not have these properties.”
Purdue Pharma Post Marketing Requirement Briefing Document, September 2020[8]
“There was not clear evidence that OxyContin’s reformulation caused heroin-naïve individuals to initiate use…”
FDA Briefing document, September 2020[9]
2021
FDA continues to encourage the development of abuse-deterrent opioids
“The FDA is encouraging the development of prescription opioids with abuse-deterrent formulations (ADFs) to help combat the opioid crisis. The agency recognizes that abuse-deterrent opioids are not abuse- or addiction-proof but are a step toward products that may help reduce abuse.”
FDA, as of March 8, 2021[10]
December 2023
FDA again confirms its approval of OxyContin[11]
Neither FDA Nor Purdue Anticipated Level of Abuse That Occurred
FDA and Purdue expected that any abuse of OxyContin would be similar to the experience with Purdue’s long-acting morphine medication, MS Contin, which has been on the market since 1987.
1995
OxyContin’s original 1995 label disclosed risk of addiction: “Delayed absorption as provided by OxyContin tablets, is believed to reduce the abuse liability of a drug.”
2002
FDA’s John K. Jenkins testified before U.S. Senate:
“At the time of approval, FDA also considered the abuse potential of OxyContin and determined that its abuse potential was similar to that of other Schedule II narcotics and we did not foresee the widespread abuse and misuse of OxyContin that has been reported in the past few years. Despite these troubling reports, however, FDA continues to believe that the benefits of OxyContin outweigh its risks when the drug is used according to the approved labeling.”
“In part, FDA based its judgment of the abuse potential for OxyContin on the prior marketing history of a similar product, MS-Contin, a controlled-release formulation of morphine that had been marketed in the U.S. by Purdue Pharma without significant reports of abuse and misuse for many years.”[2]
2019
FDA said:
“There was no evidence to suggest at the time that crushing the controlled-release capsule followed by oral ingestion or snorting would become widespread and lead to a high level of abuse.”[12]
2020
FDA continues to recognize opioid analgesics taken as prescribed rarely cause addiction:
“According to the National Institutes of Health, studies have shown that properly managed medical use of opioid analgesic compounds (taken exactly as prescribed) is safe, can manage pain effectively, and rarely causes addiction.”[13]
2001 Prescription Opioids, & OxyContin in Particular, Are Subjected to an Intense National Health Review
The need to preserve OxyContin’s important role in pain management was established, understood and stressed.
“Concerns of addiction should not prevent patients with appropriate pain conditions from using OxyContin or other narcotics for pain relief.”
OxyContin Q&A, JULY 26, 2001[14]
That same year, the FDA approved an updated OxyContin label, which was further strengthened to include language surrounding “misuse, abuse and diversion of opioids” and also added a Black Box Warning, the highest level of warning to physicians.
In addition, Purdue sent letters to 800,000 physicians nationwide informing them of the label change.[15]
2020 (January) Letter to Senate from FDA Director of Center for Drug Evaluation & Research Janet Woodcock Asserting Propriety of OxyContin Approval Process:
- “FDA has examined the original 1995 approval of OxyContin, the approvals of the supplemental applications for the 80-mg and 160-mg strengths, and the approval of the 2001 safety labeling change. All applicable statutes and regulations were followed.”
- “Approval was based on efficacy findings from six controlled studies.”
- “… when OxyContin was approved in 1995 it was not limited to the treatment of acute pain or otherwise limited in its duration of use. Chronic or long-term use (in appropriate situations), with no maximum duration, was always part of the approved use of OxyContin.”
- “The disturbing reality that there are individuals who will intentionally prescribe and dispense opioids inappropriately for financial gain underscores the complex factors that contribute to the opioid crisis.”
- “OxyContin is not appropriate for ‘as-needed dosing’ (PRN), or in the immediate post-operative period if pain is mild or not expected to persist for an extended period of time.”
- “FDA also recently funded a research project to study the effect of opioid analgesic deprescribing (i.e., tapering and/or discontinuing) on patient outcomes, including suicidality, completed suicide, and overdose…”
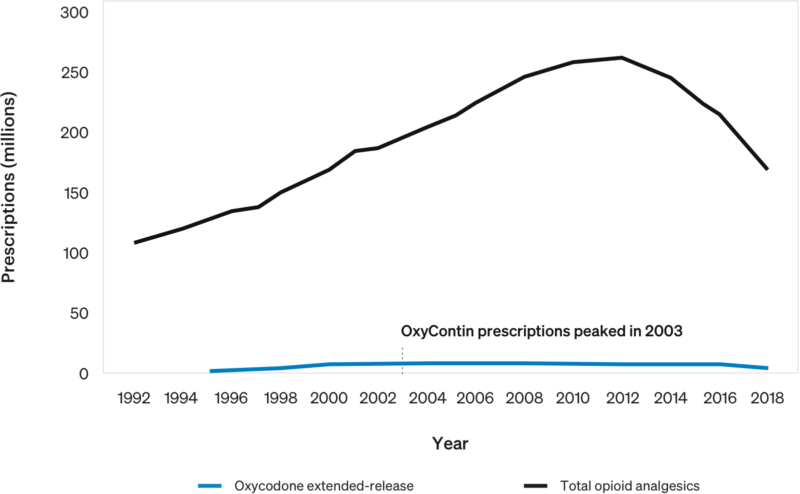
Estimated Number of Prescriptions Dispensed for Opioid Analgesics vs. Oxycodone Extended-Release from U.S. Outpatient Pharmacies[7]
False Claims of Misconduct During FDA Approval Process Were Investigated by DOJ Twice – No Action Was Taken
FDA first approved OxyContin in 1995. The Department of Justice has twice investigated the approval process of OxyContin, in 2006–2007 and a decade later in 2016–2018, including specifically regarding statements in the original label and launch materials, the underlying clinical studies, and the independence of the approval process.
- Purdue’s submissions to the FDA for OxyContin’s approval were comprehensive and in compliance with all rules and regulations.
- Purdue developed OxyContin for several years, with clinical trials beginning years prior to approval.
- FDA commented that Purdue’s application for OxyContin was “unusually robust.”[16]
- FDA approval of OxyContin’s New Drug Application (NDA) was based on six controlled clinical trials that showed the medicine is “safe and effective” – significantly more than the industry standard of two clinical trials.
- Three of the six clinical trials were for cancer pain; the others were for conditions such as osteoarthritis, low back pain and post-operative pain. (More than 700 patients participated in the trials.)
- FDA constantly monitors and evaluates the safety of OxyContin based on available science.
Purdue’s clinical trials for OxyContin were later used internally at the FDA to train new medical officers on the proper design and conduct of analgesic clinical trials.
Purdue’s trials were described as the “gold standard.”[16]
False Claim
Purdue defrauded the FDA by not making the FDA aware of a change in the package insert of a competitor’s drug.
Fact
Information about the competitor’s product, which was used as a comparative tool in Purdue’s clinical studies, was publicly known and immaterial to the FDA’s review of OxyContin. Regardless, Purdue brought the information to the FDA’s attention before it began marketing OxyContin.
False Claim
FDA official was improperly involved in the extensive approval process that determined OxyContin is safe and effective.
Fact
- Dr. Curtis Wright had an impeccable service record with the FDA and received several awards, including for Distinguished Service.
- FDA’s team consisted of at least three medical review representatives – as well as additional experts in the areas of biopharmaceutics, pharmacology and toxicology, chemistry, statistics and consumer safety.
- All of Purdue’s interactions with FDA, including formal and informal interactions with Dr. Wright, were logged in contact reports – and summaries were submitted to the FDA on a regular basis.
- During sworn testimony, Dr. Wright explained the process that eventually led to him being hired at Purdue.[17]
- Purdue’s hiring of Dr. Wright was legal, proper and transparent and consistent with industry norms.
